Drug development is one of the most expensive and time-consuming processes in modern industry. It takes, on average, ten to fifteen years and $2.6 billion to bring a single new drug from initial discovery to market approval. The attrition rate is brutal: roughly 90% of drug candidates that enter clinical trials fail. These are not new statistics. They have been the reality of pharmaceutical R&D for decades, and despite enormous investment, the fundamental timeline has barely compressed.
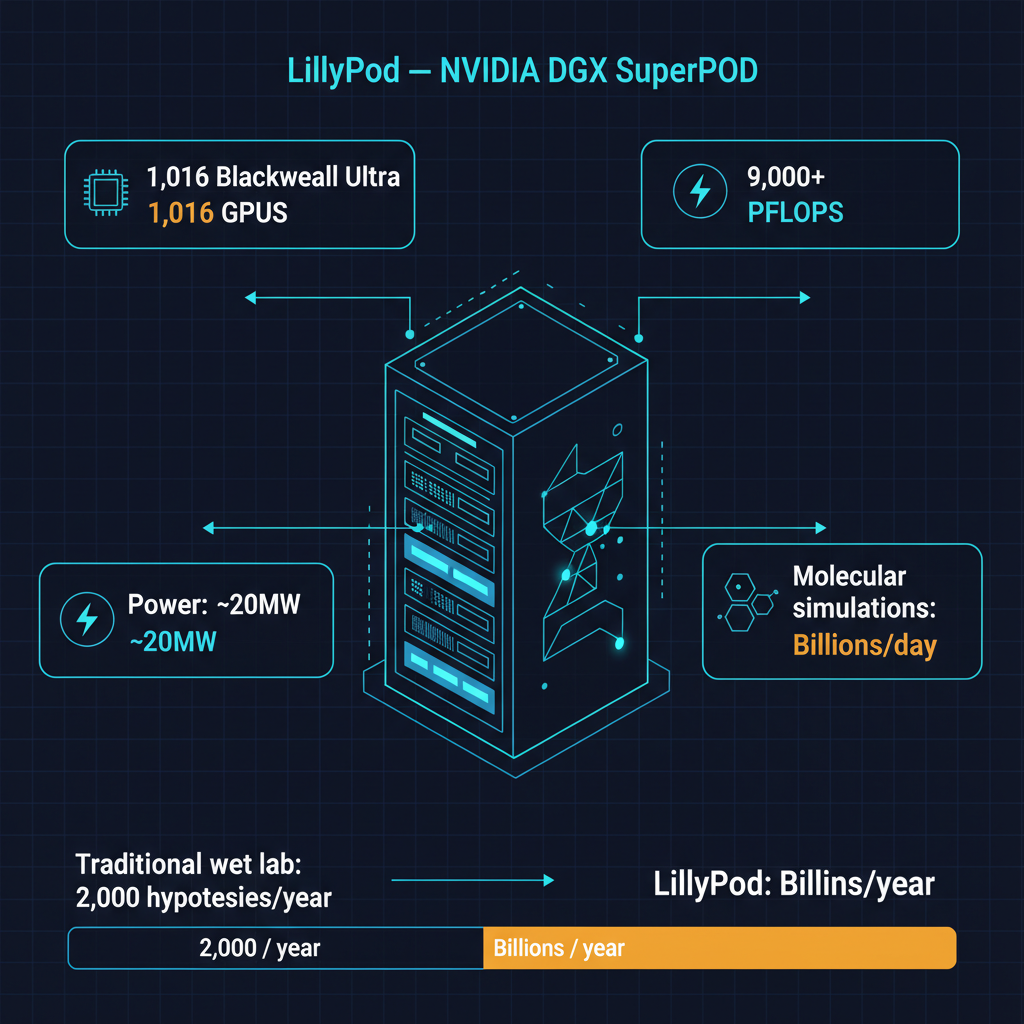
Eli Lilly just made the most significant infrastructure bet in pharmaceutical history to change this. LillyPod, inaugurated this month at the company's research campus, is built on NVIDIA's DGX SuperPOD architecture and houses 1,016 Blackwell Ultra GPUs. It delivers over 9,000 petaflops of AI compute — making it, by a considerable margin, the most powerful AI supercomputer owned and operated by a pharmaceutical company.
The numbers are impressive. What matters more is what Eli Lilly intends to do with them.
The Bottleneck LillyPod Is Designed to Break
The traditional drug discovery process begins with a target — a protein, enzyme, or receptor that plays a role in a disease. Researchers then search for molecules that interact with that target in a therapeutically useful way. This search is conducted through a combination of computational screening and physical experiments in wet laboratories.
A well-equipped wet lab can test approximately 2,000 molecular hypotheses per year. Each test involves synthesizing the molecule, preparing biological assays, running the experiments, and analyzing the results. This process is labor-intensive, expensive, and inherently serial — you can only test as many molecules as your lab capacity allows.
LillyPod is designed to invert this constraint. Using AI models trained on molecular dynamics, protein folding, binding affinity prediction, and pharmacokinetic simulation, the system can evaluate billions of molecular candidates in parallel. Instead of testing 2,000 molecules per year in a physical lab, researchers can computationally screen billions of candidates, identify the most promising ones, and then send only the highest-probability candidates to wet lab validation.
This is not a theoretical capability. The underlying AI models — descendants of AlphaFold for protein structure prediction, graph neural networks for molecular property prediction, and generative models for novel molecule design — have matured to the point where their predictions are accurate enough to meaningfully narrow the search space. LillyPod provides the compute infrastructure to run these models at pharmaceutical scale.
Why This Scale Matters
It is worth understanding why Eli Lilly chose to build at this specific scale rather than renting GPU capacity from cloud providers.
Pharmaceutical AI workloads have characteristics that make cloud computing suboptimal. The datasets involved — molecular structures, protein dynamics simulations, patient outcome data — are among the most sensitive and regulated in any industry. Moving these datasets to a public cloud introduces compliance complexity and security risk that a company like Eli Lilly is not willing to accept.
More fundamentally, the compute requirements are sustained rather than bursty. AI-driven drug discovery is not a one-time training run that requires a burst of GPU capacity. It is a continuous workflow: new molecular candidates are generated, screened, refined, and re-screened in iterative cycles that run for months or years per drug program. Owning the infrastructure makes economic sense when utilization is consistently high.
The choice of 1,016 Blackwell Ultra GPUs is also significant. NVIDIA's Blackwell architecture delivers roughly 2.5 times the AI training performance and 5 times the inference performance of the prior Hopper generation, at the same power envelope. This means LillyPod can run larger models, process more candidates per cycle, and do so with energy efficiency that would have been impossible with prior hardware generations.
What LillyPod Actually Does
Eli Lilly has outlined several specific AI workloads that LillyPod will support.
Molecular generation and screening. Generative AI models propose novel molecular structures that are predicted to interact with specific biological targets. LillyPod screens these candidates against multiple criteria simultaneously: binding affinity, selectivity, toxicity, metabolic stability, and synthetic accessibility. The ability to evaluate all of these properties computationally before synthesizing a single molecule dramatically reduces the cost and time of the discovery phase.
Protein dynamics simulation. Static protein structures, like those predicted by AlphaFold, are valuable but incomplete. Proteins in the body are dynamic — they flex, rotate, and change conformation. Understanding how a drug candidate interacts with a protein requires simulating these dynamics, which is computationally intensive. LillyPod enables molecular dynamics simulations at timescales and resolutions that were previously impractical.
Clinical trial optimization. AI models can analyze historical trial data to predict which patient populations are most likely to respond to a drug candidate, optimize trial design to detect efficacy signals earlier, and identify biomarkers that predict response. These analyses require training on large-scale patient data and running inference across millions of simulated trial scenarios.
Pharmacokinetic and toxicity prediction. Before a drug candidate enters clinical trials, researchers need to predict how it will be absorbed, distributed, metabolized, and excreted by the human body, and whether it will produce toxic effects. AI models trained on historical pharmacokinetic data can make these predictions with increasing accuracy, reducing the number of candidates that fail in expensive late-stage clinical trials.
The Broader Industry Shift
Eli Lilly is not the only pharmaceutical company investing in AI infrastructure, but they are doing it at a scale that sets a new benchmark. Other major pharma companies — Pfizer, Roche, Novartis, AstraZeneca — have built AI teams and invested in AI capabilities, but typically through partnerships with cloud providers, collaborations with AI startups, or more modest on-premises deployments.
LillyPod represents a different strategic bet: that AI-driven drug discovery is sufficiently central to the company's future that it warrants dedicated, owned infrastructure at supercomputer scale. This is not an experiment or a pilot program. It is a strategic commitment to AI as a core capability of pharmaceutical R&D.
The timing is also noteworthy. Eli Lilly's market capitalization growth over the past two years has been driven largely by GLP-1 receptor agonist drugs — the class that includes their diabetes and obesity treatments. The revenue from these blockbuster drugs provides the financial runway to make long-term infrastructure investments like LillyPod. In essence, Lilly is using the profits from its current blockbusters to build the infrastructure for discovering the next generation of drugs.
My Assessment: Promise and Honest Limitations
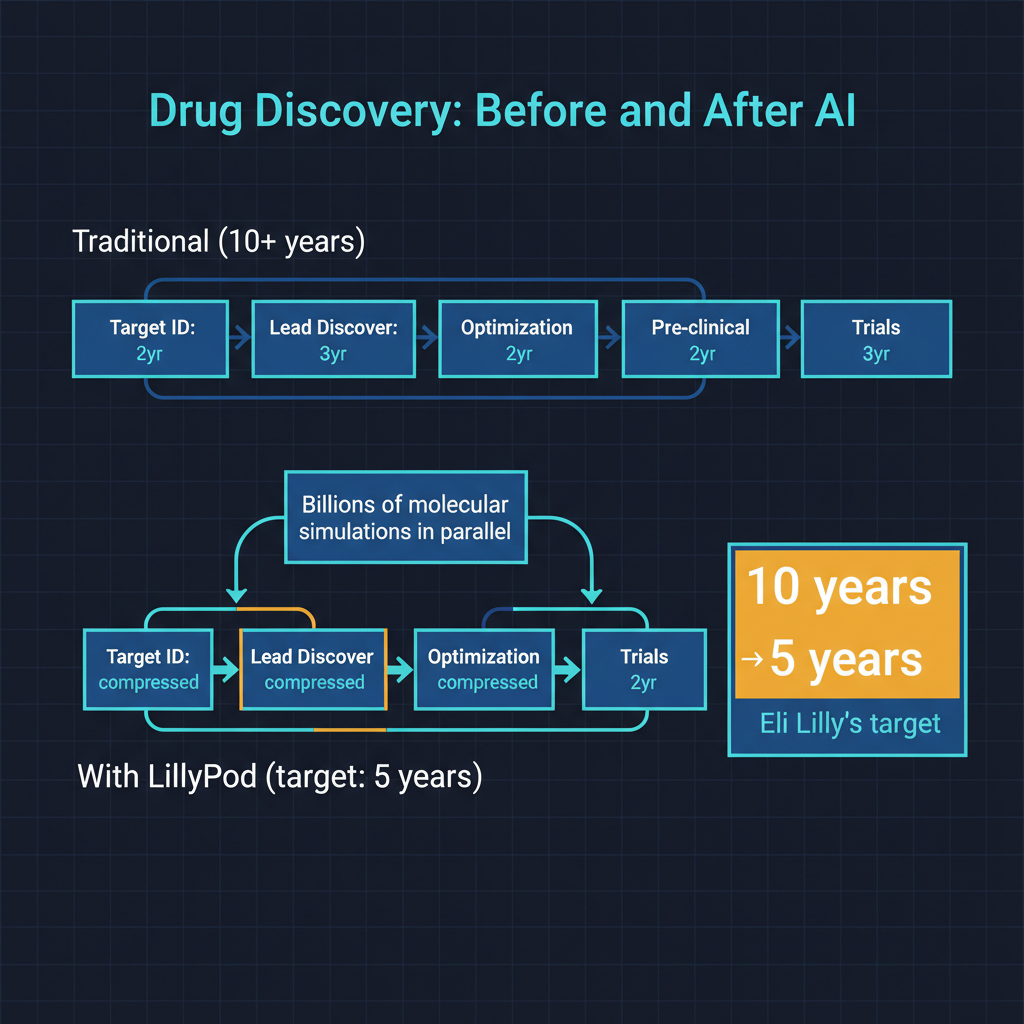
I find Eli Lilly's claim that LillyPod could cut the drug development timeline in half to be ambitious but not implausible — with important caveats.
The discovery phase of drug development, where AI has the most direct impact, typically accounts for three to five years of the total timeline. If LillyPod enables Lilly to identify high-quality drug candidates in months rather than years, the total timeline compression could be substantial. But discovery is only one phase. Preclinical testing, clinical trials (Phases I through III), and regulatory review account for the remaining years, and these phases are constrained by biology and regulation, not compute.
AI can help optimize clinical trials and improve regulatory submissions, but it cannot accelerate the biological timelines. A Phase III clinical trial that needs to demonstrate long-term safety over two years cannot be compressed by running more GPUs. The FDA's review process has its own institutional timeline. These are not computational bottlenecks that more hardware can solve.
What I find most significant about LillyPod is not the specific timeline compression claims but the strategic signal it sends. When the largest pharmaceutical companies in the world start building supercomputers, it means they have concluded that AI is not a supporting technology for drug development — it is a foundational one. The compute-intensive, data-driven, iteratively optimized workflow that LillyPod enables represents a fundamental change in how drugs are discovered.
The pharmaceutical industry has been one of the slowest sectors to adopt new technologies historically. If Eli Lilly's bet pays off — if LillyPod delivers drugs to patients faster and at lower cost — it will not just validate the investment in AI infrastructure. It will demonstrate that AI can transform even the most complex, regulated, and capital-intensive industries in the economy. That is a signal that extends well beyond pharma.



